 Metals. Ferrous metals. Iron, Steel and cast iron. Siderurgy
Metals. Ferrous metals. Iron, Steel and cast iron. Siderurgy
Prehistory
In the metal ages there were three distinct periods, each of which is named for the materials that, because of their importance at these times, represented each period:
The Copper Age (5000 years ago): owing to its low resistance, it was only used in decorative utensils.
The Bronze Age (3000 years ago): a mix of copper and tin which had better properties than either of the two metals separately.
The Iron Age (2000 years ago): much more resistant than bronze. The Hittites were the first to master iron.
 The first metal object made by Humans was invented about 7000 years ago. The first metal they used was copper. However, it wasn’t very strong so they found out that when they added tin to the copper, the mixture was stronger.
The first metal object made by Humans was invented about 7000 years ago. The first metal they used was copper. However, it wasn’t very strong so they found out that when they added tin to the copper, the mixture was stronger.
Later on, iron came and new objects become possible, such as weapons , the wheel or the plow
The wheel was very important in transport as it was used in carts pulled by bullocks.
The plow allowed humans to work faster and in greater areas
Human beings realized that raw materials were very important for making new tools. The search for these new metals and raw materials was the beginning of trade.
The general properties of metals.
- Tenacity: The ability to support impacts without breaking. Metals are tenacious.
- Ductility: The ability to form threads.
- Malleability: The ability to form sheets.
- Hardness: Scratch resistance. Metals are relatively hard materials.
- Density: the quotient of the mass and volume of a form.
- Electrical and thermal conductivity: Metals conduct heat and electricity well.
- Metallic shine: Metals characteristically shine after being recently cut.
- Except mercury all metals are solid at room temperature.
- These properties are partly due to the way the atoms are joined together in metals. Let’s see the inside of a metal.
Metallic bond
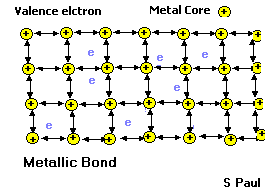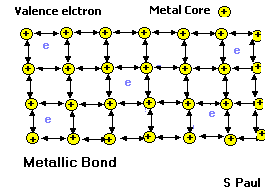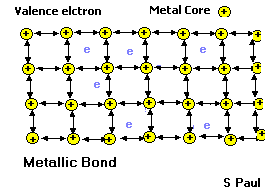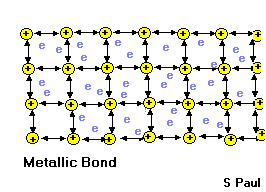
In a metal, the outer electrons are shared among all the atoms in the solid. Each atom gives up its outer electrons and becomes slightly positively charged. The negatively charged electrons hold the metal atoms together. Since the electrons are free to move, they lead to good thermal and electrical conductivity
Classification of metals.
According to their composition:
Pure metals: Metals whose crystalline structure is composed of only one metallic element. For example: gold, silver, iron, copper etc.
Metal alloys: These are a combination of one or two metals. For example: bronze, tin, steel etc
According to their iron content:
Ferrous: These are iron (the pure metal) or alloys that contain iron (steels and cast iron).
Non ferrous: These are pure metals which aren’t iron (e.g. copper) or metal alloys that don’t contain iron e.g. bronze. .
According to their density:
Ultralight: Density less than 2kg/l. For example, magnesium.
Light: Density between 2 and 5 kg. Aluminium and titanium are light.
Heavy: Density superior to 5kg/l. All the other metals are heavy.
The process of obtaining metals
1.-Obtaining the metals from mines and pits etc.
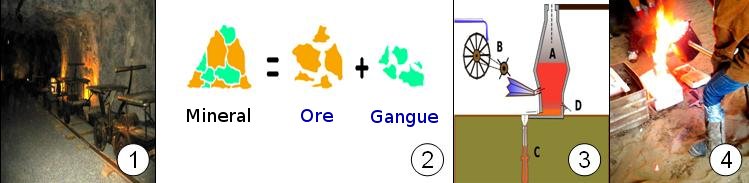
2.- Separation of the mineral into ore and gangue. The ore is the mineral that contains the metal we are interested in (for example, cuprite is the ore of copper). The gangue (impurities) is of no interest (sand, mud, other elements) .
3.- Obtaining the pure metal from the mineral (reduction process).
4.- Final treatment: They better the properties of and/or change the physical form of the metals.
Mining
Mining is an industrial activity that consists in extracting elements, compounds and minerals, which are of some economic interest, from the earth. These extractions take place in mines, pits or quarries.
When we have the mineral the next thing we must do is obtain the pure metal. The most important processes for doing this are through combustion and electrolysis
Siderurgy
Siderurgy is the term used for the industry dedicated to obtaining iron. The iron industry has its own name because of the enormity of the use of iron in society today. From iron we obtain steel, which is one of the most important materials we have.
The siderurgic process takes place in installations called ‘blast furnaces’. The raw materials used are iron (Fe), the flux (CaCO3) and carbon (C).
Siderurgic processes.
The raw materials are put into the top of the blast furnace (the throat)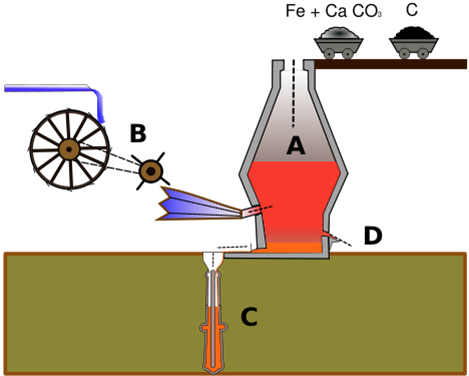
Hot air is introduced into the lower part.
When the materials descend a chemical reaction takes place. The iron separates from the elements which were contained in the original mineral.
The waste materials stay at the top and the molten metal comes out of the bottom (iron + carbon + impurities).
The molten metal is directed towards the steel industry to adjust the carbon content and the content of the other alloy materials to improve the metals properties.
Ferrous products

The ferrous products obtained after the operations realised in the blast furnace and the steelworks are classified according to their carbon content (raw material in the blast furnace). That is:
Soft iron
C < 0.1 %
It’s a brittle material with many bad mechanical properties. It has good magnetic qualities.
Used to create electromagnets, transformers and electrical appliances.
Steels
0.1% < C < 2%
Ductile, malleable, tenacious, they rust easily and they are harder the higher the % of C, they can be soldered and forged.
They are used in car bodies (see picture), in construction elements, in metal sheeting, materials made of tin and cutting instruments etc.
Cast iron
2% < C < 5%
Less ductile than steel but harder because carbon makes the metal harder but, at the same time, more fragile. It can be melted and moulded easily.
They are used in making unusually shaped pieces and bedplates for machinery (they absorb vibrations well.)
Manhole covers are an example of cast iron.
Electrolysis
This is a process principally used to obtain and/or purify non-ferrous metals. The purification or refining consists in eliminating all the impurities still left in the metal after the reduction of the mineral. It is used to obtain copper, aluminium, magnesium, zinc and titanium, among others. The term comes from the words electro – electricity and lysis – break i.e. breaking because electricity is passed through. In the case of aluminium, electrolytic refinement is called the Hall process.
Bauxite is used as a starting raw material to obtain aluminium. Using the Hall process we can get aluminium which is 99.9 % pure.
Electrolysis – the process
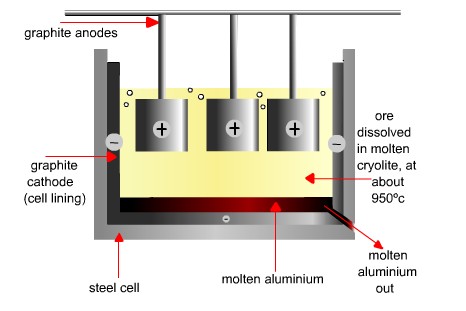
1. The bauxite ( aluminium oxide mixed with impurities ), is extracted from the Earth.
It is then treated with alkali, to remove the impurities.
The aluminium is then moved to huge tanks. In the tank there is a graphite bar, this acts as the cathode. Also there are graphite bars in the middle of the tank that act as anodes.
The aluminium is dissolved in molten cryolite. The cryolite lowers the melting point, which reduces the energy used in this process.
Electricity is passed form cathode to anode bars.
The aluminium is taken out at the bottom
Activity
1º What metals are a car made out of?. Search in the internet what metals are used in a car and answer the questions for each material
a ) Properties of the metal
b) Prices
c ) Ecological properties
2º Why are car radiators made of metal?
3º Why Aluminium Wheels? . Advantage of aluminium versus iron.
Transformer: is a device that transfers electrical energy from one circuit ( Primary ) to another ( Secondary )through the transformer’s coils.
Foul Sewer: A type of underground carriage system, for transporting sewage from houses to treatment.
Threads: See image on the left

